Research Article - (2024) Volume 13, Issue 1
Osteoarthritis, the most common joint disease worldwide, refers to a heterogeneous group of conditions that share symptoms and signs of compromised articular cartilage integrity and alterations in bone and joint margins. The pathophysiology of osteoarthritis is not comprehensively understood and its treatment is mainly symptomatic. Intra-articular injections of corticosteroids and hyaluronic acid-based preparations are commonly employed to mitigate pain and enhance joint function by replenishing damaged synovial fluid. This study aimed to evaluate the microrheological properties of the synovial fluid under conditions that mimic in vitro the healthy and the oxidized condition, which characterizes the synovial fluid in osteoarthritic subjects. Additionally, the microviscosity properties of the synovial fluid were evaluated after administration of two pharmaceutical agents for the treatment of osteoarthritis, namely the corticosteroid triamcinolone acetonide and a cross-linked hyaluronic acid. The analysis was carried out using the multiple particle technique, a type of functional microscopy that is widely used to study the structural rearrangements and the viscoelastic properties of different types of inorganic and organic fluids, which revealed that triamcinolone acetonide protects the synovial fluid from oxidation without affecting synovial fluid microviscosity. Contrarily, hyaluronic acid acts as a viscosupplement, improving the microviscosity of synovial fluid. However it proved to be ineffective against synovial fluid oxidation. These results highlighted that corticosteroids and hyaluronic acid-based formulations exert different effects on oxidised synovial fluid, and that, therefore, their administration should be chosen on a case-by-case basis.
Osteoarthritis • Synovial fluid • Microrheology • Triamcinolone acetonide • Hyaluronic acid • Oxidation • Multiple particle technique
OA: Osteoarthritis; ROS: Reactive Oxygen Species; IL: Interleukin; TNF-α: Tumor Necrosis Factor-α; NSAIDs: Non-Steroidal Anti-Inflammatory Drugs; TTA: Triamcinolone Acetate; HA: Hyaluronic Acid; SF: Synovial Fluid; MPT: Multiple Tracking Technique; MSD: Mean Square-Displacement; GAGs: Glycosaminoglycans; PBS: Phosphate Buffer Saline; AAPH: 2,2′-Azobis(2-Methylpropionamidine) Dihydrochloride
Osteoarthritis (OA) is the most common form of arthritis affecting around 237 million people worldwid. It is a progressive degenerative disorder caused by hyperostosis and degeneration of articular cartilage. Clinical manifestations include join pain, frictional sounds during movement, limitation of joint activity, joint swelling, and joint deformity. Although OA can affect any synovial joint, knee OA represents the greatest social burden [1].
The pathophysiology of OA is not well understood. Many risk factors associated with the development of the disease have been identified, including genetic and constitutional factors, and preceding joint defects or damage. In addition to mechanical disorders of the joint, inflammation, and subsequent oxidative stress, due to the production of Reactive Oxygen Species (ROS), play a key role in the pathogenesis and progression of OA [2]. Indeed, several inflammatory mediators, including Interleukin-1β (IL-1β), Tumor Necrosis Factor-α (TNF-α), and Interleukin-6 (IL-6), are up-regulated in joints affected by OA. This stimulates the production of ROS and the expression of matrix-degrading proteases, thus contributing to the degradation of structural biomolecules and joint dysfunction [3].
At present there is not a definitive cure for OA. A great effort of research has been devoted to the search for drugs capable of alleviating the symptoms, as well as to the development of pharmacological strategies to slow down or even halt further development of the disease. Indeed, it must be borne in mind that patients with OA are often old and have other comorbidities that must be taken into account when prescribing a treatment.
Drugs commonly used to alleviate the symptoms of OA include selective Cyclooxygenase (COX)-2 inhibitors, Non-Steroidal Anti Inflammatory Drugs (NSAIDs), corticosteroids and opioids. Intra articular injection of drugs for the treatment of OA is currently an option for patients who have not responded to oral or topical treatments, as this type of administration allows increasing the bioavailability of drugs at the target site, overcoming their systemic side effects.
Some randomized trials have shown that, in patients with OA, the intra-articular injection of anti-inflammatory drugs, such as methotrexate, diclofenac, and Triamcinolone Acetate (TTA), reduced pain and improved joint function for up to 3 weeks. Other studies, however, showed that, compared to other systemic non-steroidal and anti-inflammatory treatments, steroids did not produce additional benefits, especially in terms of restoration of the function [4].
Various formulations composed of cross-linked and non-cross linked fractions of Hyaluronic Acid (HA), the glycosaminoglycan that represents the main component of the Synovial Fluid (SF), are also currently available on the market as viscosupplements for the treatment of OA [5]. These preparations aim to partially restore the rheological properties of the SF and prevent further joint erosion. HA preparations should also produce pain relief and improve joint mobility.
Although the intra-articular injection of corticosteroids and HA based preparations is currently used in daily clinical practice, clinical guidelines do not recommend them as first choice drugs for the treatment of osteoarthritis [6]. Indeed, the scientific evidence on the efficacy of these products is still controversial and, in some cases, incomplete [7].
One of the aims of this study was to investigate whether there is a difference in the rheological properties of human knee SF samples under conditions that mimic healthy and OA disease conditions in vitro, the latter produced by oxidizing the SF samples with AAPH, an azo compound widely used as a free radical generator. In addition, we aimed to provide scientific evidence of the effect on the viscoelastic properties of SF of two marketed injectable preparations used for the local treatment of OA: a formulation of the corticosteroid Triamcinolone Acetonide (TTA; Kenacort 40™) and a medium molecular weight cross-linked Hyaluronic Acid formulation (HA; JonexaTM). The characterization of the rheological properties of the SF samples was carried out using the Multiple Tracking Technique (MPT), a passive microrheological method, validated in our laboratory for very small sample volumes, that is widely used to assess the elastic and viscous properties of complex fluid samples [8-10]. Briefly, fluorescent nanobeads are immersed in a fluid whose mechanical properties are extracted from the Brownian displacements of the probes, measured using digital video microscopy. Nanobeads exhibit larger displacements when their local environment is less rigid or less viscous. The amplitude of the motions over different time scales is measured by the Mean Square-Displacement (MSD) of the nanobeads. The use of a large collection of probes allows not only high temporal and spatial sensitivity, but also better statistics and easier analysis of sample heterogeneity.
We hypothesised that oxidised SF samples, which mimic the OA condition in vitro, would have a reduced microviscosity compared with control samples and, that the treatment with the two drugs used for the local treatment of OA would improve the synovial fluid microviscosity. To our knowledge, this is the first study directly comparing the effect of two different intra-articular injectable preparations for the treatment of knee OA on the viscoelastic characteristics of knee synovial fluid.
Chemicals
Kenacort 40™ (Bristol Myers Squibb, New York, NY, USA) and Jonexa Hyalastan SGL80™ (Genzyme Corporation, Pleasant View Terrace, Ridgefield, NJ, USA) were commercially available at the local pharmacy. Kenacort 40™ contains 40 mg/mL Triamcinolone Acetonile (TTA) suspended in a physiological solution. According to the manufacturer, Jonexa Hyalastan SGL80™ is a mixture of Hyaluronic Acid (HA) cross-linked with divinylsulfone and sodium hyaluronate fluid suspended into a physiological solution. Table 1 presents information on the two preparations, as reported in the package leaflets. If not explicitly stated, all chemicals and materials used in this work were purchased from Sigma Aldrich, Milan, Italy.
Synovial fluid samples
Human synovial knee fluid, was purchased from articular engineering (Northbrook, USA). The samples were collected from three female deceased donors, aged from 66 to 75 years old, not affected by OA within 48 h from death caused by intracranial hemorrhage, ovarian cancer, leukemia and ischemic stroke, respectively. The characteristics of the patients whose synovial fluid samples were analysed in this study are summarised in Table S1. Immediately upon arrival, samples were aliquoted into 200 µl vials and their biochemical and viscoelastic properties were immediately measured. Remaining aliquots were stored at -80°C until further analysis. Cycles of thawing and freezing of the samples were avoided.
Biochemical analysis of synovial fluid samples
The total protein concentration of the four samples was determined using the Bradford protein assay colorimetric kit (Bio Rad, Hercules, CA, USA) according to the manufacturer’s instructions, with bovine serum albumin used as the standard. The concentrations of sulphated and unsulphated Glycosaminoglycans (GAGs) were determined colorimetrically [11]. In this method, the concentration of total GAGs is measured by staining an aliquot of the sample by Alcian blue binding under alkaline conditions, and the sulphated GAGs by Alcian blue binding under acidic conditions. The concentration of each of the complexes is determined measuring the absorbance of each complex at 480 nm, using HA and chondroitin sulphate as standards for total and sulphated GAG, respectively. The concentration of non-sulphated GAGs (mainly hyaluronate) is obtained by difference.
Synovial fluid treatment and oxidation with AAPH
The microrheological properties of the synovial fluid were analysed in samples composed of 7.5 µl SF and 12.5 µl physiological saline (0.9% NaCl). To determine the effect of TTA and HA, 0.8 µl of the commercial products (92.1 mM TTA or 10.5 mg/ml HA) or PBS (in mM: 137 NaCl, 2.7 KCl, 1.2 CaCl2, 1.44 Na2HPO4, 1.76 KH2PO4, pH 7.9) used as control, were added.
Oxidation of the SF samples was induced by incubation at 37°C for 1 hour with 2,2′-Azobis(2-Methylpropionamidine) Dihydrochloride (AAPH). To determine the concentration of AAPH with maximal oxidant capability, the microrheological parameters were evaluated at increasing concentrations of the oxidising agent (in mM: 0 (PBS), 1.25, 2.5, 5 and 10). In each condition tested, the AAPH added to the solution did not exceed 5% of the total volume (Table 1).
| Active substance | Triamcinolone Acetonile (TTA) | Hyaluronic Acid (HA) (Cross-linked + unmodified) |
| Formulation name | Kenacort 40™ | Jonexa hyalastan SGL-80™ |
| Concentration | 40 mg/ml | 10.5 mg/mL |
| Drug’s class | Corticosteroid | Glycosaminoglycan |
| Used as | Anti-inflammatory | Visco-suplementation |
| Inactive ingredients | Benzyl alcohol, carmellose sodium and polysorbate 80 | 2.2 mg/mL Na2HPO4.7 H2O;0.26 mg/mL NaH2 PO4.H2O |
| Suggested posology | 5 to 60 mg/day | 4 mL+4 mL, 15 days apart |
Table 1. Information on commercial formulations of TTA and HA reported in their package leaflets.
Evaluation of the microrheological properties of the synovial fluid samples
Multiple Particle Tracking (MPT) was used to evaluate the microrheological properties of the SF samples in different experimental preparations. The method has been validated by measuring the microviscosity of glycerol solutions with known microviscosity. In MPT, the time course of the position of the beads in suspension inside the medium to be studied is recorded. A 20 µL aliquot of synovial fluid sample preparation and 1 µL of solution containing 200 nm diameter yellow/green fluorescent polystyrene carboxylated beads (λexc=488 nm, λem=505-515 nm; Life Technologies, Monza, Italy) were mixed, and 8 µL of the preparation, containing ≤ 100 beads/field, was deposited between two glass cover slips, and the borders were sealed to avoid evaporation. After an equilibration at room temperature for 20 min, beads were focused on the mid-height of the sample to exclude beads that might be interacting with the cover slips with a 60 × (N.A. =1.42) objective connected to a CCD video camera. Images of 1280 × 960 pixels were captured at a rate of 5 frames/s. The trajectory of the Brownian motion of the beads was recorded using the multitracker plug-in of ImageJ. About 400 beads were tracked in four to eight fields per sample. The movement of the fluorescent beads within the mucus in a given time interval, τ, is described by its mean squared displacement,
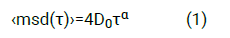
Where: α is the elastic contribution of the fluid whose value is 0<α ≤ 1. The microviscosity, η, is calculated from the Stokes-Einstein equation, as:
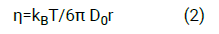
Where: kB is the Boltzmann constant; T is the absolute temperature; r is the radius of the microspheres.
Statistical analysis
Data are expressed as mean ± standard deviation. For each examined condition, samples were measured at least three times. The Kruskal-Wallis non-parametric analysis of variance followed by Dunn’s post hoc test were used to compare data sets. A value of p<0.05 was considered statistically significant. All statistical and MPT analysis was done with Igor Pro 9 software (Lake Oswego, OR, USA).
Biochemical characterization of the synovial fluid samples
The four SF samples, referred to as SF1, SF2, SF3 and SF4, showed no significant difference in total protein concentration (p=0.165). Total GAG concentration was significantly lower in SF1 and SF3 samples compared to the other two SF samples (SF2: p=0.012 and p=0.04, SF4: p=0.023 and p=0.034, respectively). No significant difference was found in the concentration of sulphated GAG among the groups (p=0.307). The amount of non-sulphated GAG, obtained as the difference between total and sulphated GAG and presumably consisting mostly of HA, was 3.41 ± 0.25 mg/ml (n=9), 3.65 ± 0.16 mg/ml (n=9), 3.12 ± 0.32 mg/ml (n=9) and 3.86 ± 0.11 mg/ml (n=9) mg/ml, respectively. Thus, the non-sulphated GAG content of SF1 and SF3 samples was lower than that of the other two samples (SF2: p=0.009 and 0.012, SF4: p=0.007 and 0.022, respectively). The concentrations of total protein and total sulphated and non-sulphated GAGs are reported in Table S2 together with the results of the statistical analysis.
Microrheological analysis of the synovial samples
The microrheological properties of the four SF samples were analysed using the MPT. The Microviscosity (η) of the four SF samples, as calculated from the plot of the mean square displacement against the time interval, was 34.79 ± 5.67 cPoise (n=5), 92.54 ± 7.91 cPoise (n=5), 26.11± 9.39 cPoise (n=5) and 137.68 ± 8.76 cPoise (n=7), in SF1, SF2, SF3 and SF4, respectively. As shown in Figure 1 had a significantly higher η compared to that of the other two SF samples (SF2: p=0.004 and 0.002, SF4: p=0.019 and 0.036, respectively. On the contrary, the elastic component of the SF, expressed by the coefficient α, and representing the non-linearity of the mean square displacement of the beads, was not statistically different in the four SF samples (1.02 ± 0.16, n=5; 0.86 ± 0.19, n=5; 0.86 ± 0.22, n=5, 0.99 ± 0.16, n=7, respectively).
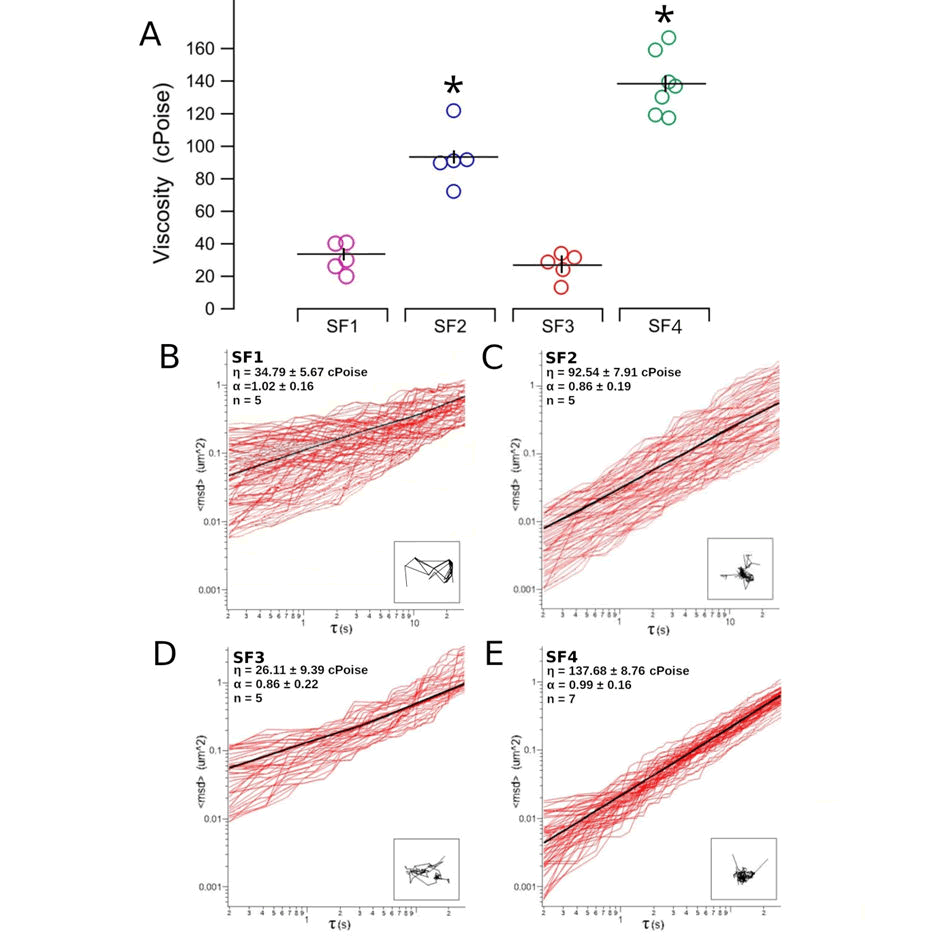
Figure 1. Microrheology of the four SF samples measured by the MPT. (A): Microviscosity of the four synovial fluid samples. The circles represent single measures (n=5, 5, 5 and 7 for SF1, SF2, SF3 and SF4, respectively), while the black horizontal and vertical lines the mean and the standard deviation of each sample, respectively. Statistical comparison of the data was made by the Kruskal-Wallis non-parametric analysis of variance followed by Dunn’s post hoc test. Asterisks indicate a statistical significance (p<0.05) from SF1. (B-E): Representative plots of the mean square displacement,
To analyse the effect of the pharmacological treatment with TTA and HA on the microrheological properties of the synovial fluid, 0.8 µl Kenacort 40™ (TTA), Jonexa Hyalastan SGL80™ (JNX) or PBS (control condition) together with 7.5 µl of the synovial samples was solubilized into 12.5 µl of a physiological solution. This determined a decrease of the microviscosity of the knee synovial samples that resulted 11.678 ± 0.44 cPoise (n=5), 33.41 ± 1.09 cPoise (n=3), 7.99 ± 0.22 cPoise (n=5), and 39.77 ± 0.71 cPoise (n=5), respectively.
As the differences in microviscosity among the four knee synovial samples were statistically significant (Figure 1), to allow comparison, for each SF sample, the microviscosity measured in each experimental condition was normalised to the mean value of the microviscosity measured in the untreated control.
Determination of the AAPH concentration with maximal effect
To determine the optimal concentration of the oxidative treatment, the SF samples were incubated at increasing concentrations of AAPH, and the effect of the treatment on the microviscosity was measured. The normalised microviscosity values of the four SF samples were plotted against the AAPH concentration and fitted with a rectangular parabola (3). As shown in Figure 2, the maximum reduction of the normalised microviscosity is 0.11, and half of the maximum effect is obtained at an AAPH concentration of 5.15 mM. Accordingly, the 5 mM AAPH concentration was chosen for the oxidative treatment in the subsequent experiments.
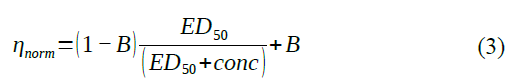
Where: ηnorm is the normalised microviscosity; ED50 is the effective concentration that produces the half of the maximum effect; conc is the AAPH concentration; B is the maximum effect on the microviscosity.
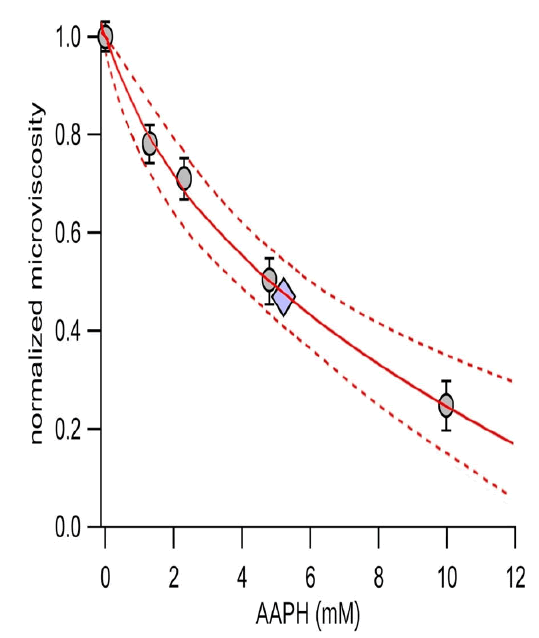
Figure 2. Effect of AAPH on the microviscosity of SF. SF samples were incubated with a physiological solution containing 0, 1.25, 2.5, 5, and 10 mM AAPH at 37°C for 1 h. Grey circles represent the normalised microviscosity (grey circles) at each AAPH concentration tested, expressed as mean ± sem. For each condition, 3 to 5 replicate measurements were made with each SF sample. The solid line represents the fit with a rectangular parabola, and the broken lines represent the 95%confidence interval of the fitting. The diamond shows the position of the half-maximal effect.
Effect of the pharmacological treatments on the microviscosity of oxidised synovial fluid
As shown by Figure 3, under control conditions, without the addition any pharmacological agent, the normalised microviscosity of SF samples treated with 5 mM of the oxidising agent AAPH is significantly lower (0.54 ± 0.16, n=13) than that of control samples not treated with AAPH (1.00 ± 0.09, n=18).
The microviscosity of SF samples supplemented with TTA in the absence of AAPH is not significantly different from that of the control (0.96 ± 0.21, n=13), indicating that TTA does not play a direct role in microviscosity determination. In contrast, when SF is treated with 5 mM AAPH in the presence of TTA, a sort of "protection" against oxidation is observed, as the microviscosity values are not statistically different from the control (1.05 ± 0.16; n=14).
In contrast, HA supplementation significantly increased the microviscosity of the SF samples (4.39 ± 0.19; n=15) with respect to the untreated control samples. But treatment with 5 mM AAPH in the presence of HA produced a significant reduction in SF microviscosity (2.06 ± 0.13; n=14). Representative plots of the mean square displacement,
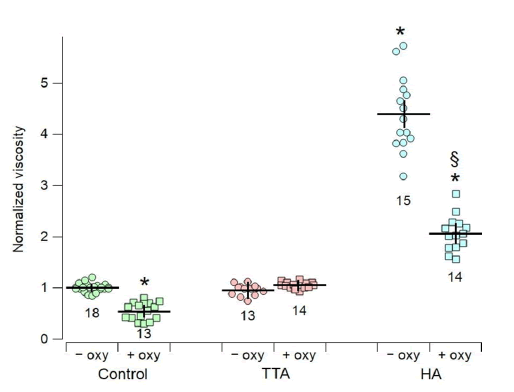
Figure 3. Normalised microviscosity obtained in SF samples incubated at 37°C for 1 h with 0 mM (-oxy; circles) or 5 mM AAPH (+oxy; squares). Treatment was carried out in the presence of PBS (Control; green), TTA (pink) or HA (light blue). In each experimental condition, microviscosity was measured in the four SF samples (5 to 7 replicates of each sample), and normalised to the mean value of the microviscosity measured in the untreated controls of each SF sample (Control, -oxy). The mean and the standard deviation are represented by the horizontal and vertical black lines in each experimental group, respectively, the number of data in each group is indicated. Asterisks (*) indicate data that were statistically different (p<0.05) from the untreated and unoxidised (-oxy) control condition, the section sign (§) indicates statistical differences (p<0.05) between the oxidised (+oxy) and unoxidised (-oxy) HA-treated preparations.
In Osteoarthritic (OA) joint, damage to the articular cartilage is caused by changes in the rheological properties of the Synovial Fluid (SF), the thin layer of fluid that acts as a lubricant on the surfaces of the articular cartilage. The SF becomes less viscous and therefore less effective in lubricating and protecting the joint. In the injured joint, the changes in the physico-chemical properties of the SF are accompanied by synovial inflammation, oxidative stress, chondrocyte apoptosis, extracellular matrix degradation, subchondral bone sclerosis and osteophyte formation [12].
The present study aimed to quantitatively evaluate the viscoelastic properties of human SF under conditions that mimic in vitro the healthy and the oxidized condition that characterizes the OA joint environment. In addition, as intra-articular injections of corticosteroids or HA-based formulations, known as viscosupplementation, are commonly used treatments of OA, aimed to minimize pain and maintain or even improve joint mobility, we also analysed the effect of their administration on SF microviscosity.
To achieve our aims, we used four human knee SF samples obtained post mortem from 4 female non OA donors. The choice to use SF from deceaded female donors, aged between 66 and 75 years, was due to the high inter-person variability of the mechanical characteristic of the SF. In fact, even in absence of a specific joint pathology, gender, age and joint workload deeply affect either the composition and the viscoelastic properties of SF [13].
As proteins and Glycosaminoglycans (GAGs) are the main components of the SF, we first determined the content of these macromolecules in the four SF samples [14]. The total protein concentration was similar in all the SF samples (~25 mg/ml). This result is consistent with those obtained by other groups. The concentration of sulphated and non-sulphated GAGs was measured by the Alcian blue colourimetric method. As reported elsewhere [15], our results showed that the concentration of the sulphated GAGs was very low, and was approximately constant in the all the samples. In contrast, the non-sulphated GAGs, constisting mainly of HA, represented the largest fraction of the GAGs. It is interesting to note that the content of non-sulphated GAG was different in the four samples. In fact, their concentration decreased following the order SF4>SF2>SF1>SF3.
To evaluate the viscoelastic properties of the four SF samples, we used the MPT, which has been previously validated in our laboratory. The advantage of this passive microrheology technique is that MPT allows the use of very small volumes (on the order of microlitres) to evaluate the viscous and elastic properties of a fluid, ensuring high spatial and temporal sensitivity [16]. Our measurements showed that the four SF samples had a statistically different microviscosity (Figure 1). Inter-individual differences in SF viscosity have been observed by other researchers and have been correlated with various factors, such as normal ageing process, gender, genetic background, obesity, previous trauma and joint injury. In contrast, the elasticity of the four samples did not differ significantly. These observations seem to suggest that in healthy elderly individuals the viscous character of SF prevails over the elastic one. The loss of elasticity of SF could be attributed to physiological processes affecting the elderly and could partly explain the increased vulnerability of their joints to compressive, tensile and shear forces that occur during daily joint movement. As observed in other studies, the difference in the microviscosity of the four samples (SF4>SF2>SF1>SF3) seemed to correlate with the content of non-sulphated GAGs, the major component of which is HA. On the contrary, no apparent correlation was observable between the content of either sulphated GAGs or proteins and the microviscosity of the samples [17].
The lubricating capacity of HA is significantly reduced in OA. This is due to a decrease in molecular size (partial depolymerisation) and HA concentration, which results in a deterioration of the elastic and viscous properties of SF [18]. As mentioned above, the intra-articular injection of corticosteroids or hyaluronic acid-based preparations are two of the most common non-surgical treatments for OA, known as viscosupplementation. The main aim of these treatments is to reduce severe pain and to try to maintain or even improve joint mobility by restoring the rheological properties of healthy SF. We have used the MPT to evaluate in vitro the effect of the administration of two injectable OA therapeutics, a corticosteroid, Triamcinolone Acetonyl (TAA; Kenacort 40™), and an HA-based viscosupplement, (Jonexa Hyalastan SGL80™), on the rheological properties of the four SF samples, previously incubated with the oxidising agent AAPH to mimic in vitro the oxidative conditions typical of the OA joint environment. Indeed, oxidative stress and the release of Reactive Oxygen Species (ROS) amplify the inflammatory state of the joint and are deeply implicated in the pathogenesis of OA. In particular, ROS interfere with the synthesis and contribute to the degradation of the extracellular matrix, which, together with synovial inflammation, lead to the joint degeneration that characterises OA.
Accordingly, the SF samples were incubated with 5 mM AAPH, which was the concentration at which half of the maximum microviscosity reduction in SF samples was obtained (Figure 2). It is important to note that treatment with the two drugs resulted in a dilution of the SF samples. The rationale for this dilution is based on the fact that the volume of the intra-articular injection of the drug used to treat OA is relatively large (Table 1) with respect to the small volume of SF present in an OA knee joint (~3. ml) [19].
As shown in Figure 3, in all SF samples, TTA acted as a protective agent against oxidation. In fact, the normalised microviscosity of non-oxidised and AAPH-oxidised TTA samples did not differ statistically from the control samples. Evidently, in vivo and in the presence of cells, the anti-inflammatory capacity of TTA certainly decreases under oxidative conditions of SF, reducing ROS formation [20]. Our results, obtained in vitro and in the absence of cells, indicate an effect of TTA per se, possibly acting as a quencher of SF oxidation.
Due to HA intrinsic high viscosity, the HA-treated SF samples showed a significant increase of their microviscosity. However, HA proved to be ineffective in protecting against AAPH-mediated oxidation, as shown by the significant reduction of the microviscosity of HA-treated SF samples incubated with AAPH. Thus, the beneficial effects of HA are likely to be exclusively due to their ability to vicariate the lubricating capacity of SF in the days immediately successive to their intra-articular administration.
Although this study has some limitations, mainly due to the small sample size, results obtained indicate that there are differences in the macromolecular content and in the rheological behaviour of SF obtained from healthy individuals within the same sex and age range. The main differences regard the content of non-sulphated GAGs and the microviscosity of the SF, which macroscopically are closely related to the capability of SF to function as joint lubricant and shock absorber. Furthermore, the results from this study confirm that corticosteroids and HA-based formulations, which are commonly administrated intra-articularly in damaged joints, have a different impact on oxidized SF, the former being protective and the latter ineffective. Finally, MPT, a technique that has been successfully used to assess the rheological features of small volumes of fluids, can also be usefully applied to analyse the microrheological properties of SF. We are confident that the results of this study will provide new information that could be useful in selecting the most effective drug for the treatment of OA based on its ability to affect joint lubrication.
This work was partially supported by Innovation and Medical Science Division, Aboca SpA Societa Agricola, Loc. Aboca 20, 52037 Sansepolcro, AR, Italy.
D.B. and O.M. designed the study. D.B. and A.L. conducted the experiments. D.B. and O.M. analysed and interpreted the results. D.B and O.M wrote the manuscript. All authors have read and agreed to the published version of the manuscript.
The datasets used and analysed during the current study are available from the corresponding author on reasonable request.
The authors declare no conflict of interest.
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
Citation: Ludovico A, et al. "Investigating the Microrheological Properties of Knee Synovial Fluid by Means of the Multiple Tracking Technique". J Arthritis, 2024, 13(1), 1-6.
Received: 16-Oct-2023, Manuscript No. JAHS-23-117001; Editor assigned: 18-Oct-2023, Pre QC No. JAHS-23-117001 (PQ); Reviewed: 01-Nov-2023, QC No. JAHS-23-117001; Revised: 14-Jan-2024, Manuscript No. JAHS-23-117001 (R); Published: 18-Jan-2024, DOI: 10.35248/2167-7921.24.13(1).105
Copyright: © 2024 Ludovico A, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.