Case Series - (2020) Volume 11, Issue 6
Background: Cerebral venous sinus thrombosis (CVST) is a rare form of stroke which still carries significant morbidity and mortality. Previous treatment regimen pertained to primarily medical therapies, with use of anticoagulants like Coumadin and Heparin. However the advent of Direct Thrombin Inhibitors (DTI) which do not need regular monitoring of blood coagulation panel and has better safety profile than Coumadin, has been suggested as a treatment modality for CVST in recent literature.
Case Description: It is a case series of 7 adult patients presenting with intracerebral hemorrhage secondary to CVST from occlusion of dural venous sinuses. Dabigatran, a DTI was used as the primary treatment modality for a period of 6 months for 5 patients who had identifiable underlying cause and lifelong for 2 patient who had no identifiable underlying cause for dural sinus thrombosis. Patients has been followed for a total period of six months while on DTI treatment, with Magnetic Resonance Venography (MRV) of the brain performed at 6 months follow up visit to assess recanalization of the obstructed dural sinuses. All the 7 patients had improved clinical outcome at 6 months follow up visit with no added morbidity upon using DTI. Five patients had complete recanalization of obstructed venous sinuses, while 2 patient had partial recanalization at 6 months follow-up visit. No patient had increased hematoma volume while on treatment with DTI.
Conclusion: This case series suggests DTI are probably effective primary treatment modality for patients with CVST with good safety profile.
Cerebral venous sinus thrombosis • Intracerebral hemorrhage • Direct thrombin inhibitors • Dabigatran • Recanalization of dural sinuses
Cerebral venous sinus thrombosis (CVST) is a rare disease with highly variable symptoms and clinical course which was once considered an entity with high morbidity and mortality. During the past decade, increases awareness of this diagnosis, improved neuroimaging techniques, earlier diagnosis after the onset of symptoms and more effective treatments like anticoagulants have improved the prognosis. Several studies attest to the fact that over 80 percent of patients with CVST now have a good neurological outcome [1-3]. The largest international multicenter trial of CVST in 2004 included very few patients from Asia and Africa, hence leaving the need for more studies on CVST from Asia especially in the age of modern neuroimaging techniques and effective anticoagulant therapies. In this regard, Chaturbedi et al. performed a retrospective observational study of 45 adult patients with CVST confirmed by radiography and fully investigated for pro-thrombotic states in a premier neuroscience institute in Nepal [4]. Anticoagulants, mostly drug Coumadin was prescribed for a total period of 6 months with transient risk factor for venous thrombosis and indefinitely in patients with confirmed thrombophilia as per the guidelines of European Association of Neurosurgeons (EFNS) [5]. In nutshell, the study showed excellent prognosis with good clinical outcome in over 85% of patients who had prompt diagnosis of CVST and introduction of effective anticoagulant therapy.
Dabigatran, a drug belonging to a new class of Direct Thrombin inhibitors appears promising and appealing as an alternative to current anticoagulants like Coumadin or low molecular weight Heparin, used in the treatment of CVST [6]. It is an oral drug, with primarily renal clearance, has shown to be as effective as Warfarin for its anticoagulant effect, with decreased risk of untoward bleeding as compared to Warfarin [7]. Hence it as a potent anticoagulant with good safety profile. In case reversal is needed, a drug called Idarucizumab is available in the market [8]. It is available in the market primarily to prevent stroke from atrial fibrillation but also has been used in patients with venous thromboembolism including CVST, albeit the data in the latter case is limited.
The author decided to use Dabigatran, which is easily available DTI in the country of this study to treat patients with CVST as a primary treatment modality.
Case Number 1
A 45 years old male who had features suggestive of sinusitis presented with 2 episodes of generalized seizure and altered sensorium. Computed Tomography (CT) scan of the head showed acute flames shaped hemorrhage in both frontal lobes with surrounding edema and mild mass effect. Patient presented with confusion, agitation and seizure.
Since the patient’s ICH had features of venous hemorrhage and history of sinusitis with fever and pain over maxillary sinuses, CVST was suspected. Noninvasive readily available diagnostic tool Magnetic Resonance Imaging (MRI) with Magnetic Resonance Venogram (MRV) of the brain with 3D reconstruction images was performed to evaluate cerebral venous sinuses thrombosis. MRV showed clearly occluded right transverse sinus secondary to thrombosis. There was also evidence of truncated anterior part of superior sagittal sinus (SSS), suggesting occlusion of SSS in its anterior third (Figure 1A) (Figure 1B).
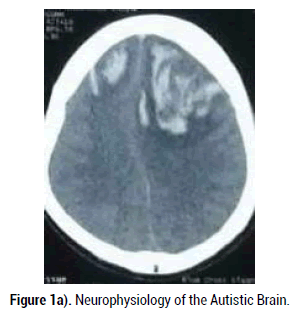
Figure 1a. Neurophysiology of the Autistic Brain.
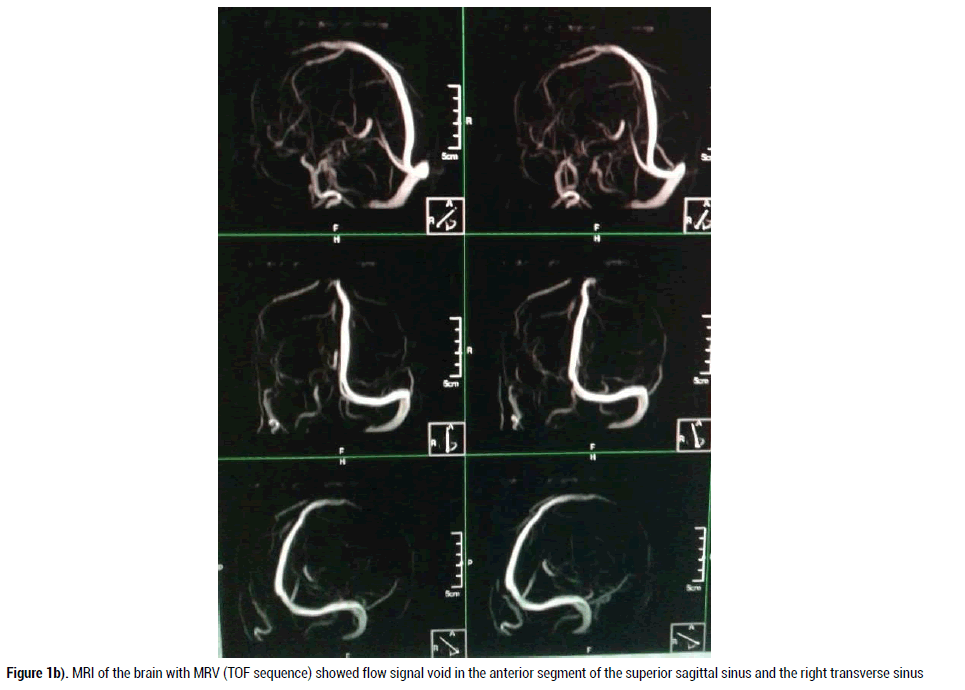
Figure 1d. MRI of the brain with MRV (TOF sequence) showed flow signal void in the anterior segment of the superior sagittal sinus and the right transverse sinus
Patient was started on Dabigatran 110 mg one tablet per oral twice a day for 6 months, apart from anti-seizure medication and appropriate antibiotics for sinusitis. Full blood coagulation profile as described under discussion section for thrombophilia screening, liver and kidney function tests were conducted prior to starting this drug, which were normal. However, D-dimer and Fibrin Degradation product (FDP) level in the blood were high, augmenting the diagnosis of venous thrombosis. Serial repeat CT brain showed resolving blood with decreasing mass effect along with improving mentation. Patient left the hospital 10 days after admission with fully intact cognition but improving motor aphasia and resolved sinusitis.
Patient was followed as an outpatient at 1, 3 and 6 months with serial repeat CT brain. He had resolved intracerebral hemorrhage (ICH) and MRI of the brain with MRV done at 6 months follow up showed patent right transverse sinus and superior sagittal sinus in its anterior part, suggesting recanalization of thrombosed venous sinuses (Figure 1C). At 6 months follow up, patient motor aphasia has almost resolved and patient had intact higher mental function.
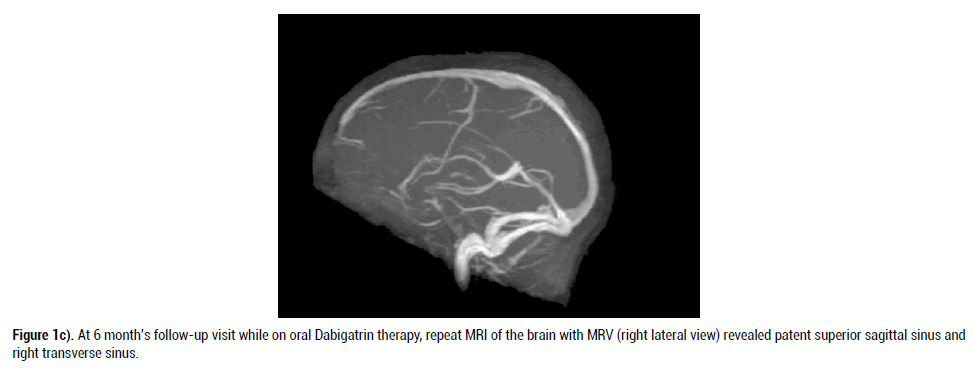
Figure 1c. At 6 month’s follow-up visit while on oral Dabigatrin therapy, repeat MRI of the brain with MRV (right lateral view) revealed patent superior sagittal sinus and right transverse sinus.
Case Number 2
A 30 years old female with 5 years history of oral contraceptive pill (OCP) use for Planned Parenthood presented to us with severe headache, left sided hemiparesis and confusion. Patient had CT scan of the head which revealed acute right parietal lobe ICH (volume: 17 mL), with perilesional edema. The hemorrhage did not follow an arterial territory and appeared as a flamed shaped hemorrhage. Taking into consideration the long term use of OCP known to cause hypercoagulable state and the characteristics of the brain hemorrhage, a MRI of the brain with MRV with 3D reconstruction was done to rule out CVST. The MRI confirmed acute nature of the bleed and surrounding perilesional cerebral edema, with no significant mass effect. MRV revealed flow signal void in the middle third of the superior sagittal sinus (SSS), suggesting thrombosed and obstructed SSS. Only about 2 cm of the SSS was occluded in its middle third segment. OCP was stopped. In retrospect, patient had confirmed left calf deep vein thrombosis (DVT) by lower extremity Doppler ultrasonography 3 years back which was treated with Coumadin for 3 months. The OCP use was considered the culprit for lower extremity DVT due to its tendency to cause hyper coagulation. Patient stopped OCP use as per doctor’s advice with recommendation for alternate contraceptive device for Planned Parenthood. Patient resumed OCP use 3 months after her DVT resolved and the drug Coumadin completed its course.
Under neurosurgical care, full panel of blood coagulation profile was tested, which came out normal. However blood D-dimer and Fibrin Degradation product (FDP) level were high. Patient did not show neurological deterioration following hospital stay. Patient was started on Dabigatran 110 mg tablet per oral twice a day, with intended treatment duration of 6 months. Serial repeat CT brain showed resolving hemorrhage and surrounding perilesional cerebral edema during her 2 weeks of hospital admission.
Patient was discharged with intact cognition, no seizures during hospital stay and with improved left sided hemiparesis, which needed support of a walker to ambulate on her own.
Patient was followed at 1, 3 and 6 months following hospital discharge. A repeat CT brain 1 month after discharge revealed completely resolved ICH. At 6 months follow-up, before considering discontinuation of Dabigatran, MRI of the brain with MRV was performed. It showed a patent SSS throughout its entire course. Dabigatran was discontinued as it ran its course and patient was discharged from neurosurgery care.
Case number 3
A 50 years old male presented to the emergency room with stupor, 1episode of seizure and 2 episodes of vomiting. An emergent CT brain showed bi-parietal lobe acute ICH (combined volume of 20 mL) with perilesional edema with moderate mass effect. There was no history of trauma, use of anticoagulant or antiplatelet drugs. Patient was admitted to ICU, intubated and kept on mechanical ventilator due to depressed level of consciousness and his inability to protect his airways. Osmotic diuresis and hyperventilation was instituted to lower ICP.
Patient did not show any hematopoietic disorder to explain the cause of the brain bleed. After patient improved clinically with no increase in the size of brain bleed, he was extubated and shifted out of ICU. He had MRI of the brain with contrast with Magnetic Resonance Angiogram (MRA) and MRV. The fact that patient had flamed shaped hemorrhage involving both parietal lobes prompted the author to think of occluded superior sagittal sinus, hence MRV was performed. The positive finding was thrombosed and obstructed SSS in its middle third part. Patient had finally a diagnosis of bi parietal ICH secondary to CVST involving middle third of SSS.
Patient was started on Dabigatran 110 mg one tablet per oral twice a day. Prior to it, a comprehensive panel to detect thrombophilia as mentioned in discussion section was carried out. These tests did not reveal any positive finding. However, blood level of D-dimer and FDP were high, augmenting diagnosis of dural sinus thrombosis. Patient had total of 2 weeks of hospital stay with serial repeat CT showing resolving hematoma and decreasing mass effect on the brain from the bleed. Patient at the time of discharge had intact cognition and no focal neurological deficit.
Like other patients, he was followed as an outpatient at 1, 3 and 6 months intervals with serial repeat CT brain, detailed neurological exam and MRI of the brain with MRV at 6 months follow-up. Patient had intact mentation but had persisting dressing apraxia, geographical agnosia, astereognosis and graph aesthesia. MRV of the brain revealed partially recanalization of SSS. It was decided that the patient needed further follow-up as he may develop isolated intracranial hypertension in the long run and a planned diagnostic catheter cerebral angiogram at 1 year after the hospital presentation to assess the patency of superior sagittal sinus. Patient has been put on lifelong Dabigatran as the choice of anticoagulant and will have further outpatient follow-up.
Following cases will be abbreviated. They all had Dabigatran as standalone treatment for CVST following confirmation radiologically. They all had elevated D-dimer and FDP. None had hematoma expansion while on DTI. All had clinical improvement at 6 months follow up as an outpatient. They were all ruled out for any underlying thrombophilia. At 6 months, radiologically 3 out of 4 patients had patent venous sinuses (previously occluded by venous thrombosis) with completely resolved ICH.
Case number 4
A 30 years old male presented with severe headache, seizure and vomiting. CT head revealed right occipital lobe ICH (volume 15mL). Antecedent history of head trauma 2 weeks back. MRI brain with MRV revealed thrombosed and occluded distal third of SSS. Dabigatran was given for a period of 6 months for the treatment of CVST with head trauma likely be the inciting factor. At 6 months follow up visit, patient had completely recanalized and patent SSS on repeat MRI brain with MRV.
Case number 5
A 45 years old chronic alcoholic with poor nutritional status and dehydration presented to us with altered sensorium, seizure and vomiting. CT head revealed left temporal lobe ICH (Volume 17 mL). MRI brain with MRV showed obstructed left transverse sinus, hence obstructing venous return from left vein of Labbe secondary to thrombosis. Dabigatran treatment was instituted for 6 months, with dehydration as the likely cause of CVST. Repeat MRV at 6 months revealed patent, hence recanalized left transverse sinus.
Case number 6
A 40 years old female presented to us with confusion, headache and speech impairment. Patient had Left frontal ICH (volume 18 mL) near primary motor cortex. MRI of the brain with MRV revealed occluded middle third of SSS, secondary to thrombosis. No underlying cause for sinus thrombosis was identified in this case. Patient was put on lifelong oral Dabigatran. Patient had no neurological complaint at 6 months follow up visit as an outpatient. MRV revealed patency suggesting recanalization of SSS.
Case number 7
A 50 years old male presented with severe headache, confusion and seizures. CT head revealed right temporal ICH (volume 15 mL) with perilesional edema. MRI brain with MRV revealed venous thrombosis and occlusion of right sigmoid sinus. Patient had history of right ear infection 1 month back which was treated appropriately with oral antibiotic. Patient was treated with Dabigatran for 6 months, with clinical improvement at 6 months follow up visit. Repeat MRV then showed partial recanalization of right sigmoid sinus but patient had no features of isolated intracranial hypertension. This patient will be followed as outpatient on a regular basis for possibility of developing delayed isolated intracranial hypertension and to evaluate the patency of partially occluded sinus radiographically.
CVST is a rather rare disease which accounts for <1% of all strokes [5]. The largest prospective multicenter international study named ISCVT shed tremendous light on the natural course and long-term prognosis of patients with CVST. With the advent of advanced diagnostic tools, better treatment modalities and keen awareness of neurosurgeons/neurologist about its existence, has significantly improved morbidity and mortality associated with CVST which was once dreaded as a life threatening disease. Dural sinuses, deep veins (like Internal Cerebral Vein) and cortical veins can be occluded by thrombosis resulting in venous infarction, ischemic or hemorrhagic. The most common dural sinus involvement is superior sagittal sinus, followed by transverse sinus. MRI of the brain with MRV is a known non-invasive tool requiring no contrast administration with Time of Flight (TOF) sequence to detect venous thrombosis [9]. The diagnosis is aided by D-dimer and Fibrin Degradation Product (FDP) as surrogate in confirming the diagnosis of venous thrombosis.
Complete blood work up with thrombophilia screening (antithrombin III deficiency, Protein C and S deficiencies, anticardiolipid antibodies, antinuclear antibody, prothrombin time and activated partial thromboplastin time) along with Human Immunodeficiency Virus (HIV) testing was performed to detect the underlying etiology in all patients. D-dimer and fibrin degradation products (FDP) blood levels were estimated as a marker of thrombosis in all the patients. These markers were evaluated at the entry of patients to the ICU, but prior to initiating anticoagulation therapy.
Majority of the patients with diagnosed CVST generally receives Unfractionated Heparin as the initial treatment, followed by Warfarin for a period of 6 months if transient risk factors are present or indefinitely for patient(s) with confirmed thrombophilia. EFNS guidelines on the treatment of CVST recommend anticoagulation for it, provided that there is no contraindication to anticoagulation. If reversible risk factor is the cause of cerebral venous thrombosis (for example infection, dehydration and oral contraceptive pill use), it should be treated or eliminated. If the cause for cerebral venous thrombosis remains unidentified, then anticoagulation can be used for a protracted course, the guideline for which is not set. It is left to the judgment of treating physician. Concomitant intracranial hemorrhage related to CVST is not a contraindication for anticoagulant therapy as per the EFNS guidelines. The Cochrane Stroke group concluded that despite limited evidence available, anticoagulant treatment for CVST appears to be safe and was associated with reduction in the risk of death or dependency [10].
Invasive procedure like use of Intra-arterial thrombolytics and mechanical thrombectomy can be used in patients with life threatening dural sinus thrombosis. However, their use is limited to dedicated stroke center and patients refractory to medical therapy.
An eye catching alternative to current types of anticoagulant therapy is newer form of anticoagulants called direct thrombin inhibitors (DTI). Available DTI include competitive direct thrombin inhibitor (e.g., oral dabigatran), reversible direct thrombin inhibitors (argatroban and bivalirudin, both intravenous only), and irreversible direct thrombin inhibitors (e.g., desirudin subcutaneously and lepirudin intravenously). Their main current indications include primary stroke prevention in patients with non-valvular Atrial Fibrillation, treatment of Venous Thromboembolism including cerebral venous sinus thrombosis (CVST), and management of heparin-induced thrombocytopenia. Data on the incidence and outcomes of direct thrombin inhibitor-related ICH is scant. Dabigatran seems to be associated with an ICH rate of 0.2–0.3 % per year, which is lower compared with warfarin. Recently, a dabigatran-specific monoclonal antibody, Idarucizumab (Praxbind®), has been approved for clinical use. The use of Idarucizumab in dabigatran reversal was demonstrated in an interim analysis of the Reversal Effects of Idarucizumab on Active Dabigatran Study (the RE-VERSE AD study).
Dabigatran has a numbers of important advantages over Warfarin and is likely to become increasingly frequently prescribed. Dabigatran is a synthetic benzamidine-derived DTI that rapidly and directly inhibits free and fibrinbound thrombin. In nutshell, it binds to thrombin and blocks its enzymatic activity. Its potentially greater efficacy, simple dosing, and better safety profile, particularly with regard to intracerebral hemorrhage, are all direct result of its appealing pharmacokinetic and pharmacodynamics profile. Its main comparative disadvantage relate to its monitoring and reversal, and any physician should be familiar with these. Dabigatran has renal clearance, its dosage needs to be adjusted in patients with impaired renal function. The best tests to check the effect of Dabigatran for its anticoagulant effect is Thrombin Clotting Time (TT) and Ecarin Clotting Time (ECT), rather than unreliable Prothrombin Time (PT) and activated partial thromboplastin time (aPTT) in case of Dabigatran use. However, they are generally not performed regularly. They are sought in case life threatening bleeding occurs while on this drug or a surgery is contemplated. As stated, Idarucizumab is available for the reversal of the anticoagulant effect of Dabigatran.
The DTI demonstrated increased utility compared with Vitamin K antagonist (VKA) like Warfarin by more predictable pharmacokinetics not requiring international normalized ratio (INR) testing or complicated dose adjustments, while at the same time having similar efficacy to VKA in treatment of acute venous thromboembolism (VTE), similar, if not better, rates of bleeding and mortality. Subsequently, increased prescribing of these medications has been seen in the last decade. However, due to the rarity of CVST, trials comparing DTI to heparin or VKA have been minimal. There have been nearly 10 years of experience with direct oral anticoagulants in the treatment of venous thrombosis and prevention of stroke in patients with atrial fibrillation, with purported efficacy and safety in comparison with heparins and vitamin K antagonists.
At present, when prospective clinical trials are missing or underway to evaluate the efficacy and safety of DTI in the treatment of CVST, this case series will lend a small but meaningful optimism towards its use. All patients in this series had hemorrhagic venous infarction, which is an ideal scenario to check for the safety of DTI. Five out of seven patients in this case series had complete recanalization of the obstructed sinus on Dabigatran use, suggesting good efficacy of this DTI. This study will add to the growing literature on the efficacy and safety profile of DVT.
Based on the finding in our case series (albeit small in sample size), Dabigatran and DTI in general appears as an appealing alternative to current categories of anticoagulants used to treat CVST. This case series suggests DTI has good efficacy to re-establish venous outflow in patients with CVST while maintaining a good safety profile. The fact that it can be taken orally without the need for regular monitoring for blood coagulation profile makes it more appealing to the treating physician.
I owe a debt of gratitude to the neuro-radiologist, hematologists and intensivist who has helped me with this study in providing insight to the study and care of the involved patients. No financial aid or grant has been received from any source to conduct this study.
Disclosure: None
Conflict of Interest: None
Citation: Abhishek Chaturbedi, Jitendra Thakur. Dabigatran, A Direct Thrombin Inhibitor (DTI) as the First Line Treatment for Cerebral Venous Thrombosis. An Updated Case Series. J Neurol Neurophysiol, 2020, 11(6), 001-004.
Received: 18-Jul-2020 Published: 13-Oct-2020
Copyright: © 2020 Abhishek C. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.