Research Article - (2025) Volume 6, Issue 1
Background/aims: Despite state-of-the-art diagnostic/therapeutic approaches, cancer accounts for the majority of mortality rate annually, and patients with breast cancer are not exception. So, a comprehensive understanding on the biological activities of Circulating Tumor Cells (CTCs) seems as a more effective cancer biomarker. This study aims to investigate the importance of detected CTCs for identification of metastatic cells in patients with breast cancer.
Materials and methods: Peripheral blood samples were collected from 11 patients with breast cancer, and 8 healthy volunteers. CD45 coupled magnetic microbeads were used to enrich CTCs. Then, the cells were exposure with fluorochrome-labeled antibodies, being cultured in an optimized condition, and subsequently analyzed by BD FACS Aria™ III Cell Sorter. Statistical analysis of correlation was done by SPSS version 21.0.
Results: No statistically-significant difference was found between results related to the tumor staging, ER, PR, Cerb-B2, CEA, CA15-3, and CTC count in patients with breast cancer (p<0.05). Meanwhile a significant correlation was determined between presence of EpCAM +CTC, and CEA levels.
Conclusion: Results of our study provide a biological tool for a better prognostication and patient stratification aimed at improving clinical outcomes in precision oncology for patients with breast cancer.
Breast cancer • Circulating tumor cells • Clinical applications • Flow cytometry • Liquid biopsy
From epidemiological aspects, cancer has been considered as one of the leading causes of mortality/morbidity rate worldwide, and Turkey is not an exception [1]. Cancer will have been considered as the first cause of mortality through a rapid increase by 2030. According to the statistics related to the cases of breast cancer, there is a swift increase in the annual mortalities in patients with breast cancer (32.371 cases by 2040), making it the most common type of cancers among Turkish woman (25,320 newly-diagnosed cases of breast cancer).
The cellular characteristics of cancer, such as uncontrolled cell proliferation, invasive spreading, and altered signaling pathways, contribute to cancerogenesis [2]. Despite advanced therapies, cancer-related deaths persist due to the prominence of metastatic phenotypes, chemoresistant cells, abnormal signaling, and tumor heterogeneity. The metastasis process involves local infiltration, intravasation, survival and transport through the bloodstream, extravasation, and colonization in distant organs [3], in which, Circulating Tumor Cells (CTCs) enter lymphatic system and/or blood circulation and play an inevitable role in the tumor progression/metastasis [4]. Literally, the description of CTCs in the peripheral blood of patients with cancer dates back to Ashworth’s observations in 1869. Then, a trial was reported regarding the CTCs levels in metastatic breast cancer [5]. Those disseminating CTCs may either passively or actively enter the bloodstream via Epithelial to Mesenchymal Transition (EMT) or centrosomes amplification, attaching to the endothelium in the target organs to invade the surrounding parenchyma and form new tumors through downregulation of the cell adhesion molecules and tight junctions (e.g. Ecadherin), leading to the stimulation of CTCs generation, CTCs survival in blood circulation, and extravasation into the secondary organ. They undergo Mesenchymal to Epithelial Transmission (MET), enabling the colonization of CTCs, converting the disseminated mesenchymal tumor cells back to the epithelial cells, and ultimately resulting in metastatic lesions, necrosis, or remaining in the dormancy situation [6]. Interestingly, only 2.5% of those CTCs cause micro-metastasis, and 0.01% of them cause macrometastasis. Therefore, the presence of CTCs in the peripheral blood of patients with breast cancer is an indicator of cancer metastasis and secondary tumor formation, because even a very small amount of CTCs (1 CTC in 10 mL blood) is an indicator of poor prognosis.
From clinical aspects, CTCs can be recruited as useful predictors of Progression-Free Survival (PFS), Overall Survival (OS), and Quality of Life (QOL), making them a promising biomarker for tumor phenotyping, mutation profiles, possibility of tumor spreading, selection of the best therapeutic approach, and monitoring the response to the treatment for patients with breast cancer [7]. As CTCs can be time-dependently found only in the serum of patients with cancer, they act more specifically than other tumor markers, and their serum levels can be dependent to the tumor staging (benign or malignant tumors) [8]. Additionally, CTCs can be used as neo-adjuvant treatment for the assessment of chemotherapy (as a prognostic marker for early relapse, and triple-negative breast cancer) through providing a “liquid biopsy” [9]. Although CTCs are rare tumor cells, they have many different subpopulations such as epithelial-origin CTCs, mesenchymal-origin CTCs, and Circulating Stem Cells (CSCs). Being responsible for the metastasis, CTCs can remain dormant for many years in the peripheral circulation. CTCs biologically are found 1 in 105-108 numbers of leukocytes in the peripheral blood of patients with cancer [10]. Special strategies for the isolation and enrichment of viable CTCs can be size-based, density-based, immunomagnetic separation, and microfluidic-based [11]. In this case, Epithelial Cell Adhesion Molecule (EpCAM) is one of the mostly-known surface molecules (transmembrane glycoproteins being originated from epithelial) on CTCs for the analysis of their molecular and functional characterization.
According to the aforementioned points, authors of this study aim to demonstrate the association among Estrogen Receptor (ER), Progesterone Receptor (PR), Cerb-B2, serum tumor markers (including CEA, and CA15-3) levels, tumor staging, and CTC levels in the serum samples of patients with breast cancer, verifying the clinical values of CTCs in clinical practice.
Study population
Participants of this study included 11 females in the patient groups, and eight females in the control group.
Study design
Participants of the study were pleased to come to the laboratory. After signing an informed written consent, antecubital venous blood samples (7.5 mL) were withdrawn by an authorized supervisor with sterile gauge needles, and then collected into vacutainer blood collection tubes (10 mL) containing anticoagulant EDTA (done at AYBU, Ankara Ataturk Training and Research Hospital, and Ankara Numune Training and Research Hospital (Ankara, Turkey)). Collection of fresh blood in the case group was done after receiving chemotherapy. Our case group included never pretreated patients with breast cancer who are newly diagnosed. Our control group included healthy volunteer participants with no history of cancers or chronic inflammatory disorders.
Study procedure
Here, enrichment of CTCs was done aimed at removing peripheral blood cells except than the CTCs at room temperature through isolation of collected Peripheral Blood Mononuclear Cells (PBMCs) by Ficoll gradient centrifugation. Then, to eliminate the leukocytes, the retained cells were coated with anti-CD45 microbeads. Magnetic positive cell population were labelled with fluorochrome-labeled antibodies (including anti-CK14+, anti-CK15+, anti-CK17+, anti-CK19+, anti-CK7+, anti-CK8+, anti-EpCAM+, and anti-CD45-, and cultured in an optimized condition. Direct immunofluorescence assay was performed using monoclonal antibodies against the cell surface markers. Sorting cells were carried out with BD FACS Aria™ III Cell Sorter (Figure 1). According to our CTC gating strategy, subpopulations such as CK+CTC, EpCAM +CTC, CK+CSC, EpCAM+CSC, and CSC were arranged for classification of CTCs.
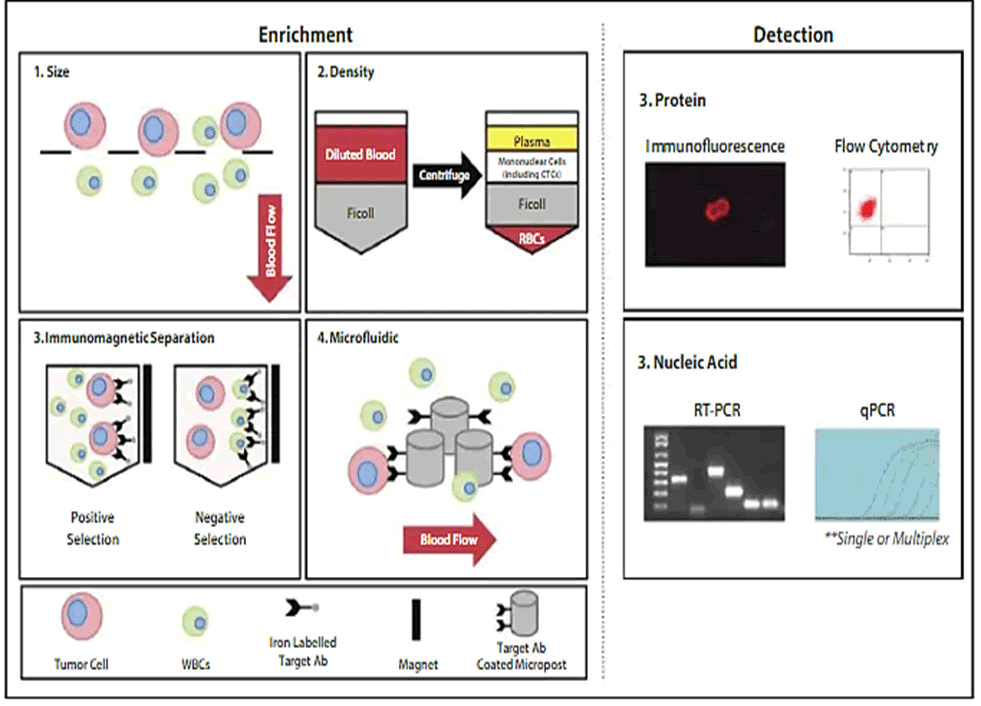
Figure 1. Cellular procedure of the study for detection and enrichment of CTCs.
Statistical analysis of the data
IBM SPSS statistics computer software for windows version 21.0 (IBM Corp. released 2012. Armonk, NY, USA) was used for statistical analysis of the acquired data. Categorical variables were expressed with percentage (%). CTC counts were expressed in median (minimum-maximum). Shapirowilk test was used for the investigation of the distribution of age and CTCs count. Mann- Whitney U test was used for the comparison of the numbers of CTCs detected in the control and case groups. Wilcoxon test was used for the investigation of cell surface markers on CTCs (CK+CTCs, EpCAM +CTCs, CK+CSCs, EpCAM+CSCs numbers obtained from CK19 based and EpCAM-based gating methods) in the case group. Poliserial correlation coefficient was used for investigation of the results from comparison of Cerb-B2, and the CTC numbers in the case group. In addition, spearman rho coefficient was used to investigate the results from comparison of other cancer serum markers, and the CTC numbers in the case group. Results were expressed as mean plus minus Standard Deviation (Mean ± SD). Statistical significance was considered at p value <0.05.
According to the Table 1, mean age of the case, and control groups were calculated 60.70 ± 17.07, and 56.90 ± 16.03 years, respectively.
| Demographic characteristics | Parameters | Number (%) |
| Age | ≤ 50 | 5 (45.5%) |
| > 50 | 6 (54.5%) | |
| Operable | Negative | 4 (57.1%) |
| Positive | 7 (42.9%) | |
| Stage | I | - |
| II | 3 (22.3%) | |
| III | 5 (45.4%) | |
| IV | 3 (22.3%) | |
| ER | Negative | 2 (18.2%) |
| Positive | 9 (81.8%) | |
| PR | Negative | 2 (18.2%) |
| Positive | 9 (81.8%) | |
| Cerb-B2 | 0 | 3 (27.2%) |
| 1+ | 4 (36.4%) | |
| 2+ | 2 (18.2%) | |
| 3+ | 2 (18.2%) |
Table 1. Demographic characteristics of our studied population.
Changing CTC and CSC values in the case group (during stage II and stage IV) after the first cycle of chemotherapy are shown in Table 2. While EpCAM-CTC and CK19-CTC were not detected in the aforementioned group, CSC was detected in some healthy individuals.
|
CTCs subpopulations |
Cell groups determined according to the gating strategy |
Cell surface antigens |
|
CK+ CTC |
CK14,15,16,19 based CTCs (CK19 based CTCs) |
CK14,15,16,19+/CK7,8+/EpCAM+/CD45- |
|
EpCAM+ CTC |
EpCAM- based CTCs |
EpCAM+/CK14,15,16,19+/CK7,8+/CD45- |
|
CK+ CSC |
CK14,15,16,19 based CSC (CK19 based CSC) |
CK14,15,16,19+/CK7,8+/ CD44+/CD24- |
|
EpCAM+ CSC |
EpCAM based CSC |
EpCAM+/ CD44+/CD24- |
|
CSC |
CSC based |
CD44+/CD24- |
Table 2. Results of CTCs subpopulation and their surface antigens.
Results of CTCs count in the case group, and control group are shown in Tables 3 and 4, respectively. According to the Table 3, CK19 +CTC and EpCAM+CTC were detected above the threshold value in 6 individuals in the patient group with CK19-based gating, and EpCAM +CTC-based gating, respectively (threshold value CTC ≥ 5). The median of the number of CK+CTC determined by the CK19-based gating strategy is 5 (minimum-maximum: 1-16) in the patient group. When the CK+CTC values of the patient group and the control group were compared, it was observed that the number of CK+CTC was significantly higher in the patient group (Table 5, Z=3.789; p<0.001).
| Patients group | The number of cells | The number of CTCs subpopulation | ||||
|---|---|---|---|---|---|---|
| CK+CTC | EpCAM+CTC | CK19+CSC | EpCAM+CSC | CSC | ||
| S2P1 | 4,13,472 | 3 | 5 | 7 | 4 | 18 |
| S2P2 | 39,25,000 | 6 | 5 | 9 | 4 | 1106 |
| S2P3 | 7,48,213 | 6 | 4 | 12 | 5 | 1587 |
| S3P1 | 8,50,770 | 6 | 7 | 10 | 5 | 2709 |
| S3P2 | 19,31,701 | 7 | 9 | - | - | - |
| S3P3 | 5,07,836 | 3 | 3 | 11 | 1 | 31 |
| S3P4 | 15,11,855 | 3 | 0 | 4 | 5 | 39 |
| S3P5 | 36,362 | 2 | 4 | 6 | 3 | 5 |
| S4P1 | 13,32,010 | 1 | 1 | - | - | - |
| S4P2 | 1,94,448 | 16 | 10 | 13 | 17 | 851 |
| S4P3 | 49,75,000 | 5 | 7 | - | - | - |
| S2P2R | 2,51,669 | 6 | 6 | 3 | 5 | 29 |
| S4P2R | 13,76,309 | 10 | 10 | 7 | 9 | 46 |
Table 3. Count of CTCs in the case group after receiving chemotherapy.
| Control group | The number of CTCs (Events) | The number of CTC subpopulation | ||||
|---|---|---|---|---|---|---|
| CK19+CTC | EPCAM+CTC | CK19+CTC | EPCAM+CTC | CSC | ||
| C1 | 3,25,935 | 0 | 0 | - | - | - |
| C2 | 1,97,459 | 0 | 0 | 1 | 0 | 7 |
| C3 | 1,69,147 | 0 | 0 | 0 | 0 | 2 |
| C4 | 3,01,548 | 0 | 0 | 0 | 0 | 0 |
| C5 | 8,20,971 | 0 | 1 | - | - | - |
| C6 | 6,56,732 | 0 | 0 | 2 | 0 | 18 |
| C7 | 6,06,663 | 0 | 0 | 1 | 0 | 1 |
| C8 | 11,28,019 | 0 | 0 | 0 | 0 | 192 |
Table 4. Count of CTCs in the control group.
|
CTC subpopulations |
The group of patients (Number of cells) |
The group of control (Number of cells) |
Z |
p |
|
CK+CTC |
5 (1-16) |
0 (0-0) |
3.789 |
<0.001 |
|
EpCAM+CTC |
5 (109) |
0 (0-1) |
3.352 |
0.001 |
|
CK+CSC |
9.5 (4-13) |
0.5 (0-2) |
3.116 |
0.001 |
|
EpCAM+CSC |
4.5 (27) |
0 (0-0) |
3.244 |
0.001 |
|
CSC |
445 (5-2709) |
4.5 (0-192) |
2.262 |
0.02 |
Table 5. Distribution of cell numbers detected in patient and control groups.
In the control group, CTCs were not obtained in this subpopulation. The median number of EpCAM+CTC detected by the EpCAM-based gating strategy was obtained as 5 (minimum-maximum: 0-10), and 0 (minimum-maximum: 0-1), respectively, in the patient and control groups. EpCAM+CTC number is higher in patient group compared to control group (Table 5, Z=3.352; p=0.001).
When the number of CK+CSC, EpCAM+CSC and CSC detected in the patient and control groups were compared, it was found that more cells were detected in the patient group (p<0.05, Table 6). The median of the detected CK+CTC and CK+CSC numbers were calculated as 2.5 (minimum-maximum: 0-16) and 5.0 (minimum-maximum: 0-13), respectively (Table 5). In line with statistical analysis, CK+CSC number was determined to be higher than CK+CTC number (Z=2.456; p=0.014; Table 6).
|
Subpopulations |
CTC |
CSC |
Z |
p |
|
CK+ |
2.5 (0-16) |
5 (104) |
2.456 |
0.014 |
|
EpCAM+ |
1.5 (109) |
2.0 (0-7) |
1.065 |
0.287 |
Table 6. Comparison of CK+CTC and EpCAM+CTC numbers with CK+CSC and EpCAM+CSC.
The median of the detected EpCAM+CTC and EpCAM+CSC numbers is 1.5 (minimum-maximum: 0-10) and 2.0 (minimummaximum: 0-7), respectively. CTC and CSCs obtained based on EpCAM were found to be similar in number (Z=1.065; p=0.287) (Table 6).
It was observed that there was no statistically significant relationship between tumor stages, ER and PR states, Cerb-B2 values, serum tumor markers (CEA, CA15-3), and CTC subpopulations determined by the TNM values in the case group (p<0.05; Table 7).
However, there were a strong positive correlation between CEA and EpCAM +CTC, a strong negative correlation between PR status and CSC number, and a moderate negative relationship between Cerb-B2 and CK19+CSC (Table 7)
| Pathological values | CTC subpopulations | ||||
| CK+CTC | EpCAM+CTC | CK+CSC | EpCAM+CSC | CSC | |
| Tumor grade1 | 0.283 | 0.191 | 0.220 | 0.325 | -0.067 |
| ER | 0.339 | 0.175 | 0.655 | 0.399 | 0.655 |
| PR | 0.000 | 0.191 | -0.267 | -0.476 | -0.802* |
| Cerb-B21 | -0.268 | -0.350 | -0.584* | 0.122 | -0.148 |
| CA153 | -0.074 | 0.348 | 0.429 | -0.221 | -0.405 |
| CEA | 0.422 | 0.78** | 0.381 | 0.196 | 0.071 |
| Note: 1Poliserial relation coefficient was calculated. Others are the Spearman rho coefficient. *p<0.05; **p<0.01 | |||||
Table 7. Relationship between tumor-related values in the patient group and CTC subpopulations.
According to the alarmingly-rapid increase in the mortality/morbidity rates of patients with breast cancer, and the distant metastatic growth, the existent diagnostic and therapeutic approaches are of low efficiency. It seems that biological activities of CTCs are considered as a promising strategy for diagnosis, treatment, prediction of early diseases relapse or cancer metastasis, detection of chemoresistance, detection of anti-cancer drug resistance, patient stratification, and cancer progression in patients with breast cancer [12]. By the usage of surface antigens, those detected CTCs have shown clinical validity and utility, making them as a dynamic prognostic factor in patients with early breast cancer [13]. In comparison with “solid/tissue biopsies”, those aforementioned clinical applications of CTCs and their real-time follow-up of tumor dynamics can be realized at any stage in the course of diseases through “liquid biopsies” which is a noninvasive (or minimallyinvasive) modality [14]. In addition, the presence of CTCs during follow-up indicated a poor prognosis especially in patients with highrisk breast cancer. Furthermore, an enhanced risk of osseous metastatic disease (or distant disease recurrence at multiple sites) can be found at patients with early breast cancer before and after adjuvant chemotherapy, leading to making an accredited clinical decision as therapeutic interventions for those patients [15].
Several studies investigated the potentials of CTCs as a tumor marker (CK+CSC, EpCAM+CSC, and CSC) for prediction of cancer development and metastasis in patients with breast cancer [16]. In our study, CTCs were detected in all of the studied patients with breast cancer, however, CK+CTC was not detected as a result of CK19-based gating in the control group, and EpCAM+CTC was detected as a result of EpCAM-based gating in only one patient. In addition, there was a statistically-significant increase in the number of CK+CTC in the case group (Z=3.789; p<0.001), when compared to the control group. Similarly, there was a statistically-significant increase in the number of detected EpCAM+CTC in the case group (Z=3.352; p=0.001) when compared to the control group. Although it was thought that there might be a relationship between the number of detected CTCs and tumor histopathology, while some studies have not approved this relationship [17-19].
In our study, no statistically-significant difference was found between the results of tumor-associated criteria (including: Staging, Cerb-B2 value, ER status, and PR status), and the number of CK+CTC, EpCAM+CTC in the case group (p<0.05). In the same way, some of examined literatures depicted no statistically-significant difference between the results of tumor-associated criteria (including: Tumor size, staging, lymph node involvement, histological grade, ER status, and PR status), and the number of CSC (CD44+/CD24-) in the patients with breast cancer [20].
Results of our study approved no statistically-significant differences between the results of tumor-associated criteria (including: Tumor stages, ER status, Cerb-B2 value), and the number of CSCs in the case group, however, there was a negative relationship between the number of CSCs, and PR status. Furthermore, in our study, there was no statistically-significant differences among the numbers of CK19 +CSC, EpCAM+CSC, CSC, and tumor-associated criteria (including: Staging, Cerb-B2 value, ER status, and PR status), however, there was found a statistically-significant decrease in the number of CSC among CTC subpopulations (p<0.05). In addition, a statisticallysignificant increase was found in the number of CK+CSC, when compared to the number of CK+CTC (Z=2.456; p=0.014). Whereas, the number of EpCAM+CTC and EpCAM+CSC were similar (Z=1.065; p=0.287) in in the case group.
It has been investigated whether CTCs can be a more reliable biological tool for cancer diagnosis and monitoring the treatment in patients with breast cancer. In this study, although there was no statistically-significant difference between CA15-3 and the number of the cells in subpopulations of CTCs, a strong positive correlation was determined only between the number of EpCAM+CTC and serum levels of CEA (p<0.05).
In this study, there was a significant decrease in the results acquired from the number of CK+CSC, as well as the number of CSC in the patients with breast cancer after receiving chemotherapy, when compared to the CTC numbers. While there was an increase in the number of EpCAM+CSC. Therefore, CTCs can be a promising tool for monitoring the treatment in patients with breast cancer.
To conclude, according to the constantly-growing mortality rate of patients with breast cancer, there is an imperative need to the more efficient approaches like CTCs as a biological marker for diagnosis, treatment, monitoring of the treatment, and cancer progression. Results of our study have approved clinical applications of CTCs as a multifunctional and indicative biomarker for identification of metastasis or cancer progression, as well as a biological tool for a better prognostication and patient stratification aimed at improving clinical outcomes in precision oncology for patients with breast cancer, based on their successful detection and enrichment through our modified method (providing a liquid biopsy). For further pre-clinical/clinical investigations, a scientific collaboration among oncologists, cancer immunobiologists, personalized medicine specialists, medical biotechnologists, cancer biochemists, medical geneticist, medical laboratory scientists, basic medical scientists, diseases-specific biomarker specialists, and health system coordinators are highly recommended.
The authors would like to thank all of the participations in this study.
Supervision, E.S.; Conceptualization and Study Design, E.S., and B.EY.; Methodology, E.S.; Search Strategy, E.S., O.OG., and B.EY.; Literature Review, E.S., and B.EY.; Academic, Scientific, and Grammatical Revision, E.S.; Preparation of the First Draft of the manuscript, E.S., and B.EY.; Preparation and Revision of the Final Draft of the Manuscript, E.S.; Visualization, image, and table designation, B.EY; and E.E.; Data Acquisiotion, E.S., A.C., and B.EY.; Validation, E.S.; Definition of Intellectual Content, E.S.; Investigation of the Clinical and Experimental Studies, E.S., and B.EY.; Formal and Statistical Data Analysis, E.S., O.OG., and A.C.
All of the authors listed on the title page attest to the fact that they have read and approved the final version of the article, and they have received an electronic copy of the manuscript. The requirements for authorship have been met, and each author believes that the manuscript represents honest work. Also, they attest to the validity and legitimacy of data, and agreed to submit this manuscript to this journal.
There are no competing interest disclosure regarding the concept, and publication of this article.
This project is financially supported by a grant from committee of scientific research projects in the AYBU (approved code: 1676).
This study accessed ethic‘s approval from Human Sciences Ethics Committee of Ankara Yildirim Beyazit University (AYBU) (no. 02.02.2015-11).
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
Citation: Yazici BE, et al. "Circulating Tumor Cells as a Liquid Biopsy Biomarker in Patients with Breast Cancer". J Cell Mol Biol Res, 2024, 6(1), 1-6.
Received: 27-Nov-2023, Manuscript No. JMCB-23-121185; Editor assigned: 29-Nov-2023, Pre QC No. JMCB-23-121185 (PQ); Reviewed: 13-Dec-2023, QC No. JMCB-23-121185; Revised: 03-Jan-2025, Manuscript No. JMCB-23-121185 (R); Published: 13-Jan-2025, DOI: 10.35248/JMCB.25.06(1).001
Copyright: © 2025 Yazici BE, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.