Journal of Multiple Sclerosis
ISSN - 2376-0389NLM - 101654564
Case Report - (2021) Volume 8, Issue 10
Multiple sclerosis is a chronic inflammatory disease of central nervous system that commonly affects young adults. Although ocular movement disturbances are commonly encountered in multiple sclerosis, bilateral horizontal gaze palsy is quite rare. We present a case of young woman presented with dizziness, intractable vomiting and visual disturbances for one week, on examination found to have complete bilateral horizontal gaze palsy while vertical gaze was intact. Brain MRI showed a small lesion in posterior part of medial pontine tegmentum bilaterally which was responsible for patient`s symptoms. She received five doses of one-gram methylprednisolone, her symptoms gradually improved over one week after treatment.
Multiple sclerosis • Bilateral horizontal gaze palsy • Paramedian pontine reticular formation • Medial longitudinal fasciculus
MRI: Magnetic Resonance Imaging
FLAIR: Fluid Attenuated Inversion Recovery
DWI: Diffusion Weighted Image
CSF: Cerebrospinal Fluid
MOG: Myelin-Oligodendrocyte Glycoprotein
AQP: Aquaporin
PPRF: Paramedian Pontine Reticular Formation
MLF: Medial Longitudinal Fasciculus
NMO: Neuromyelitis Optica
MS: Multiple Sclerosis
DMT: Disease Modifying Therapy
A 26-year-old woman, a known case of migraine and sinusitis otherwise healthy, referred from emergency to neurology for evaluation as she presented with acute onset and progressive course of severe dizziness, frequent vomiting, imbalance and visual disturbances for 1 week. She was seen by ophthalmology 5 days prior to presentation and her examination was normal. Also she was seen by otolaryngology and given vestibular suppressants but without improvement. On examination she was fully conscious, looked ill and pale, bilateral horizontal gaze was totally impaired with both eyes fixed in midline. Convergence and vertical eye movements were intact. No appreciated horizontal saccades with oculocephalic maneuver. She denied diplopia but there was visual blurring which disappears on closing either eye. She had bilateral peripheral facial weakness, other cranial nerves were intact, she had normal motor power, sensation and coordination in upper and lower limbs but she could not tandem walk and had an unsteady gait.
Brain MRI showed a small lesion in the brain stem involving the posterior part of medial pontine tegmentum bilaterally appeared hyperintense in T2WI and FLAIR sequences, with subtle restricted signal on DWI (Figure 1). Several foci of altered signal in T2WI and FLAIR noted at the periventricular white matter, centrum semiovale bilaterally and left temporal sub-cortical region, some of the lesions are oriented perpendicular to lateral ventricles (Figure 2). No appreciable post contrast enhancement.
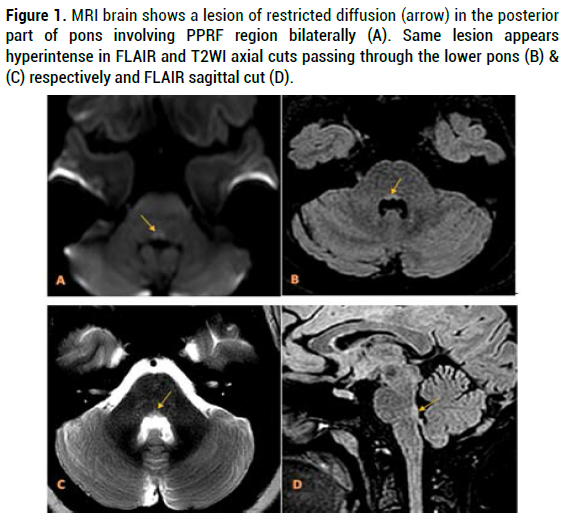
Figure 1. MRI brain shows a lesion of restricted diffusion (arrow) in the posterior part of pons involving PPRF region bilaterally (A). Same lesion appears hyperintense in FLAIR and T2WI axial cuts passing through the lower pons (B) & (C) respectively and FLAIR sagittal cut (D).

Figure 2. MRI brain T2WI axial cuts show hyperintense lesions (arrows) in left temporal subcortical region (A) and bilateral periventricular white matter (B, C & D).
Cerebrospinal fluid was within normal limits but oligoclonal IgG bands were detected in CSF but not in serum. Anti-MOG and Anti-AQP4 antibodies were negative. Other investigations included cervical and thoracic spine MRI, routine blood tests and immunology blood tests were normal.
After MRI results, methylprednisolone 1000 mg/day for 5 days was started. On 2nd day of treatment dizziness and gait improved. On 3rd day, a mild improvement in right eye abduction observed as well as facial weakness. One week after completion of pulse steroid therapy, her eye movements totally returned to normal.
Diagnosis of multiple sclerosis was made based on revised McDonald criteria 2017 (one clinical attack + dissemination in space in MRI+ demonstration of specific oligoclonal bands in CSF) [1]. On follow up, she was started on interferon B-1a weekly intramuscular injections.
Complete bilateral horizontal gaze paralysis is a rare disorder which has been reported in bilateral pontine lesions due to infarction, hemorrhage, metastasis or demyelinating lesions [2]. Bilateral horizontal gaze palsy may be caused by bilateral involvement of the paramedian pontine reticular formation (PPRF) and/or bilateral abducens nucleus, or the raphe which is affecting the descending horizontal gaze pathways [3]. Specific pathways (medial longitudinal fasciculus) and nuclei (abducens, oculomotor) are responsible for lateral gaze. The sixth nerve nucleus contains two types of neurons: motor neurons and internuclear neurons. The axons of the internuclear neurons cross to the contralateral side in the lower pons and, after ascending in the medial longitudinal fasciculus (MLF), synapse in the portion of the third nerve (oculomotor) subnucleus that innervates the medial rectus muscle. Thus, horizontal gaze involves synchronous activation of the lateral rectus muscle of one eye and the medial rectus muscle of the other, via the MLF. This final common pathway for horizontal gaze is controlled by the vestibular, optokinetic, smooth pursuit, and saccadic systems [4]. At the bedside, pontine lesions usually can be differentiated from supranuclear lesions in the frontal lobe by associated neurologic findings. The oculocephalic or doll's eye maneuver is helpful. Passive horizontal rotation of the head directly stimulates the sixth nerve nucleus via the vestibuloocular reflex and will overcome gaze palsies induced by frontal lobe lesions but will not overcome gaze palsies caused by pontine nuclear and infranuclear lesions [5].
Thus, bilateral horizontal gaze palsy in our patient can be explained by involvement of abducens nuclei and either the MLF or PPRF bilaterally. Bilateral lower motor neuron facial weakness indicated additional involvement of facial nerve nuclei or fasciculi which may label this syndrome as a variant of eight and half syndrome. Classic eight-and-ahalf syndrome is caused by a unilateral lesion in the dorsal tegmentum of the caudal pons involving the PPRF and the MLF, as well as the nucleus and/or the fasciculus of the facial nerve. It is characterized by one-anda- half syndrome (internuclear ophthalmoplegia in addition to horizontal gaze palsy) and an ipsilateral cranial nerve VII palsy (lower motor neuron-type). The affected nuclei may include either a combination of the ipsilateral seventh and sixth nerve nucleus and the ipsilateral MLF or a combination of ipsilateral seventh and ipsilateral PPRF along with the ipsilateral MLF [6].
An important differential diagnosis needed to rule out was neuromyelitis optica (NMO). In the light of presenting symptoms of nausea and persistent vomiting and the lower pontine lesion which close to the area postrema in medulla, NMO should be suspected, However, absence of anti AQP-4 and anti-MOG antibodies in serum, absence of optic neuritis and absence of long extensive transverse myelitis in cervical and thoracic spine MRI as well as presence of IgG oligoclonal bands in CSF made the possibility of NMO less likely [7]. Lastly, our patient was diagnosed with multiple sclerosis based on characteristic brain MRI demyelinating plaques, presence of oligoclonal bands in CSF (which were not simultaneously found in serum) and her clinical syndrome that was justified by a lesion in the brain MRI1 [8]. She showed rapid improvement on methylprednisolone therapy and was started on a DMT on follow-up.
Although ocular movement disorders are common in multiple sclerosis, bilateral horizontal gaze palsy is exceptionally rare. There are few case reports describe patients with similar clinical presentation finally diagnosed with MS. Understanding anatomical distribution of gaze control centers in the brain stem help localizing the lesion which further guide clinicians to suspect the underlying pathology and to order appropriate investigations that eventually lead to proper diagnosis and management.
Citation: Gamea, Mohamed Salah, AlAkkad A, Ibraheem R. Complete Bilateral Horizontal Gaze Palsy as Initial Manifestation of Multiple Sclerosis: A Case Report. J Mult Scler (Foster City), 2021, 8(10), 273.
Received: 28-Jul-2021 Published: 29-Nov-2021, DOI: 10.35248/2376-0389.21.8.273
Copyright: © 2021 Gamea MS, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.